PTC124-GD-041-DMD
(aussi appelée étude 041)
Essai clinique sur l’ataluren pour la dystrophie musculaire de Duchenne causée par une mutation non-sens (NMDMD).
L’étude 041 est un nouvel essai clinique pour les garçons et les jeunes hommes qui vivent avec la dystrophie musculaire de Duchenne causée par une mutation non-sens (nmDMD).
Qui peut participer à l’étude 041?
Afin de s’assurer que les participants qui s’inscrivent à l’étude 041 en tirent le meilleur bénéfice et pour garantir leur sécurité, des critères stricts ont été établis afin de déterminer qui peut participer à l’étude. Votre médecin vous aidera à décider si vous/votre enfant pouvez y participer.
Principaux critères d’inclusion
- Documentation prouvant qu’un test génétique a été effectué et démontre que vous/votre enfant êtes atteint de DMD.
- Vous/votre enfant êtes de sexe masculin et âgé d’au moins 5 ans.
- Vous/votre enfant êtes capable de marcher au moins 150 mètres sans aide pendant un test de marche de six minutes.
- Vous/votre enfant êtes capable de passer certains tests chronométrés.
- Vous/votre enfant avez suivi un traitement par corticostéroïdes (c.-à-d. prednisone, prednisolone ou deflazacort) pendant au moins 12 mois immédiatement avant le début du traitement à l’étude, sans changement significatif de la dose dans les 3 mois précédant le début de l’étude (autre que pour tenir compte d’un changement de poids corporel).
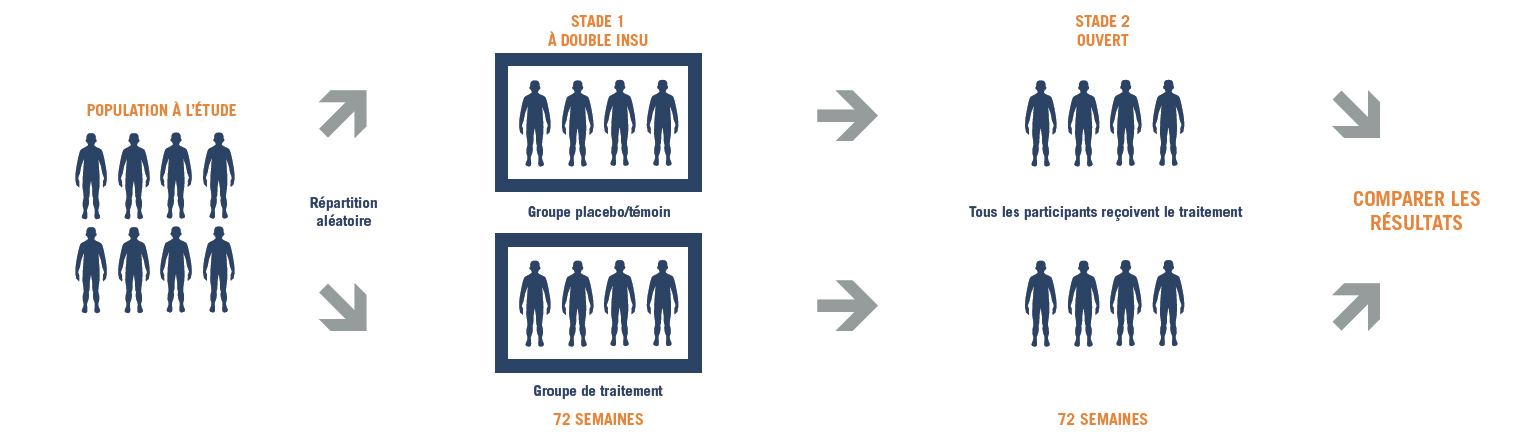
Conception de l’essai
Le stade 1 de l’étude 041
Le stade 1 de l’étude 041 examine la manière dont le médicament expérimental, l’ataluren, affecte la capacité de marcher et l’endurance chez des individus de sexe masculin âgés de 5 ans et plus atteints de nmDMD, comparativement au placebo (une substance qui a la même apparence et le même goût que l’ataluren, mais qui ne contient pas le médicament). L’étude examinera aussi la capacité des participants à monter et descendre des escaliers, marcher/courir certaines distances et utiliser leurs bras, et elle mesurera leur fonction pulmonaire. L’innocuité (sûreté) de l’ataluren sera aussi évaluée.
Le stade 2 de l’étude 041
Le stade 2 de l’étude 041 va comparer les résultats obtenus chez les participants ayant commencé à prendre l’ataluren depuis le début de l’essai et ceux obtenus chez les participants ayant commencé à prendre l’ataluren depuis le début du stade 2. Tous les participants prendront part aux deux stades de l’essai.
Si vous/votre enfant décidez de vous inscrire à l’étude 041, vous pourriez contribuer à faire émerger une meilleure compréhension de la nmDMD, ainsi qu’à mettre au point des traitements actuels et futurs, qui pourraient bénéficier aux patients atteints de nmDMD à l’avenir.
Foires aux questions
Qu’est-ce que l’ataluren?
L’ataluren est un médicament expérimental actuellement à l’étude pour le traitement d’individus atteints de nmDMD âgés de 5 ans ou plus qui sont toujours capables de marcher. Il est administré trois fois par jour, mélangé à un aliment liquide ou semi-solide et pris par voie orale. Les individus atteints de nmDMD sont incapables de produire une protéine, la dystrophine, qui aide à maintenir les muscles en bonne santé. Sans dystrophine, les muscles s’affaiblissent au cours du temps. PTC Therapeutics étudie l’ataluren pour déterminer s’il aide l’organisme à produire la dystrophine, ce qui pourrait aider à ralentir l’affaiblissement musculaire.
L’ataluren est-il sûr?
L’ataluren a été étudié chez plus de 1 000 patients dans des essais cliniques. Pendant ces essais cliniques, il est apparu que l’ataluren était bien toléré chez les patients atteints de nmDMD, et son profil d’innocuité favorable a été démontré pour cette affection.
Aurai-je des frais à payer pour participer à cet essai?
Non, tous les frais que comporte l’essai, y compris les examens physiques, la sélection, les analyses de laboratoire et autres tests ainsi que le coût du médicament, seront couverts par PTC Therapeutics, qui finance l’étude 041.
Vous pourriez être remboursé pour tous les frais raisonnables* de déplacement, de repas et d’hébergement nécessaires à vos visites au centre d’étude clinique, qui auront lieu environ toutes les 12 semaines pendant le premier stade de l’essai, puis toutes les 24 semaines pendant le deuxième stade de l’essai. * Les frais remboursés sont assujettis à certaines limites, qui vous seront expliquées quand vous viendrez à la clinique.
Comment l’ataluren va-t-il affecter les autres traitements que je prends?
Si vous/votre enfant prenez des médicaments, alors le médecin vous informera/informera votre enfant des problèmes potentiels et discutera des options de traitement avec vous/votre enfant. Toutefois, pour participer à l’essai, vous/votre enfant devez prendre des médicaments appelés corticostéroïdes, et l’ataluren peut être pris avec ces médicaments.
Que se passera-t-il si l’ataluren est approuvé dans mon pays, ou une fois qu’il le sera? Puis-je quitter l’essai?
Si vous/votre enfant vous inscrivez à l’étude 041, nous aimerions que vous/votre enfant vous engagiez à suivre jusqu’au bout les obligations liées à l’étude, à moins qu’un médecin ne détermine que vous/votre enfant devriez arrêter votre participation pour des raisons de santé ou de sécurité. Toutefois, la participation à cette étude est entièrement volontaire et si vous le souhaitez, vous/votre enfant pouvez vous retirer de l’étude en tout temps, pour toute raison, ou sans raison.
Que dois-je faire pour participer à l’étude 041?
Si vous/votre enfant souhaitez participer à l’essai, avant de vous y inscrire, vous/votre enfant allez devoir discuter de l’essai avec votre médecin et accepter (consentir) d’y participer. Vous/votre enfant devrez signer un ou des formulaires de consentement. Le ou les formulaires de consentement fournissent des informations détaillées, qui décrivent les risques et les avantages de la participation à l’essai. Si vous/votre enfant avez des questions concernant ces renseignements, parlez-en à votre médecin, car il est important que vous soyez bien informé. Le ou les formulaires de consentement expliquent aussi les faits essentiels concernant l’essai, en des termes appropriés à l’âge du lecteur. Lorsque vous/votre enfant aurez signé ce ou ces formulaires, on vous en donnera des copies à conserver.
La prochaine étape consiste en quelques vérifications pour s’assurer que vous/votre enfant êtes admissible à l’essai. Ces vérifications, appelées procédures de sélection, comprendront des tests chronométrés pour évaluer votre capacité/la capacité de votre enfant à courir/marcher 10 mètres, monter 4 marches, descendre 4 marches et vous/se mettre debout en 30 secondes à partir d’une position couchée sur le dos. Ces vérifications seront effectuées dans les deux semaines précédant le début de l’essai pour vous/votre enfant. Un test de génotypage, visant à s’assurer que vous/votre enfant êtes porteur de la mutation non-sens dans le gène de la dystrophine, sera effectué à la référence, soit au début de l’essai.
Que devrai-je faire pendant l’étude 041?
Si vous/votre enfant êtes accepté dans l’essai, vous serez ou il sera réparti aléatoirement soit dans le groupe ataluren, soit dans le groupe placebo.
L’ataluren ou le placebo se prennent par voie orale. Chaque jour pendant la durée de l’essai, vous/votre enfant devrez préparer et prendre le médicament ou le placebo trois fois par jour : le matin, le midi et le soir.
Idéalement, il faut compter un intervalle d’environ 6 heures entre les doses du matin et du midi, un intervalle d’environ 6 heures entre les doses du midi et du soir, et un intervalle d’environ 12 heures entre les doses du soir et la dose du lendemain matin.
Pour obtenir plus de renseignements
Courriel : patientinfo@ptcbio.com
Numéro sans frais : +1 (866) 282-5873
Demandez le document original : mcduberger@laforcedmd.com
Apprenez-en plus
- Apprenez-en plus sur la mutation non-sense : ici
- Apprenez-en plus sur l’ataluren (Translarna™) : www.ptcbio.com (anglais) et www.doctissimo.com (français)
- Translarna™ (ataluren)
- Les pionniers dans le traitement de la DMD